Because of the disease characteristics in DM1 and DM2, appropriate molecular testing and reporting is very important for the optimal counseling in myotonic dystrophy. Here, we describe best practice guidelinesfor clinical molecular genetic analysis and reporting in DM1 and DM2, including presymptomatic and prenatal testing.
Category Archives: General Info
Cuyamycin Molecule looks promising for Myotonic Dystrophy Treatment
A new study published shows that a small molecule (Potential new treatment) disrupted the long CTG repeats but left short repeats mostly alone.This was tested in mice that had myotonic dystrophy and seemed to work well. Here is a piece form the journal about the impact of this study
Significance
Development of small-molecule lead medicines that potently and specifically modulate RNA function is challenging. We designed a small molecule that cleaves r(CUG)exp, the RNA repeat expansion that causes myotonic dystrophy type 1. In cells and in an animal model, the small-molecule cleaver specifically recognizes the 3-dimensional structure of r(CUG)exp, cleaving it more selectively among transcripts containing short, nonpathogenic r(CUG) repeats than an oligonucleotide that recognizes RNA sequence via Watson-Crick base pairing. The small molecule broadly relieves disease-associated phenotype in a mouse model. Thus, small molecules that recognize and cleave RNA structures should be
Care recommendations for patients with myotonic dystrophy
Published in 2018 is a consensus based approach for the myotonic dystrophy patient community. This gives general guidelines on how to approach, test and intervene in patients lives to achieve the most optimum outcomes.
Care-recommendations-for-adulats-with-Myotonic-DystrophyReview Article on Myotonic Dystrophy
A new article was published on myotonic dystrophy from a number of researchers in Spain. This was a broad review by many specialists. We do not yet have a full review of the article but when the full article is available we will review it more thoroughly. Here are a few key conclusions:

The genetic diagnosis of myotonic dystrophy should quantify the number of CTG repetitions. -reason this gives some idea of the severity of the disease
Myotonic Dystrophy patients need cardiac and respiratory lifetime follow-up. These symtoms get worse with time and must be tracked
Before any surgery under general anaesthesia, a respiratory evaluation must be done for myotonic dystrophy patients. surgery is serious and the anesthesiologist needs to know the strength of the respiratory system.
Dysphagia in myotonic dystrophy must be screened periodically. – This can cause severe problems and can give an idea of the progression fo the disease
Genetic counselling must be offered to patients and relatives with myotonic dystrophy.
Here is the abstract from pubmed
Neurologia. 2019 Apr 16. pii: S0213-4853(19)30019-2. doi: 10.1016/j.nrl.2019.01.001. [Epub ahead of print]
Clinical guide for the diagnosis and follow-up of myotonic dystrophy type 1, MD1 or Steinert’s disease.
[Article in English, Spanish]Gutiérrez Gutiérrez G1, Díaz-Manera J2, Almendrote M3, Azriel S4, Eulalio Bárcena J5, Cabezudo García P6, Camacho Salas A7, Casanova Rodríguez C8, Cobo AM9, Díaz Guardiola P4, Fernández-Torrón R10, Gallano Petit MP11, García Pavía P12, Gómez Gallego M13, Gutiérrez Martínez AJ14, Jericó I15, Kapetanovic García S16, López de Munaín Arregui A17, Martorell L18, Morís de la Tassa G19, Moreno Zabaleta R20, Muñoz-Blanco JL21, Olivar Roldán J4, Pascual Pascual SI21, Peinado Peinado R22, Pérez H23, Poza Aldea JJ10, Rabasa M24, Ramos A3, Rosado Bartolomé A25, Rubio Pérez MÁ26, Urtizberea JA9, Zapata-Wainberg G27, Gutiérrez-Rivas E28.
Author information
Abstract
BACKGROUND AND OBJECTIVES:
Steinert’s disease or myotonic dystrophy type 1 (MD1), (OMIM 160900), is the most prevalent myopathy in adults. It is a multisystemic disorder with dysfunction of virtually all organs and tissues and a great phenotypical variability, which implies that it has to be addressed by different specialities with experience in the disease. The knowledge of the disease and its management has changed dramatically in recent years. This guide tries to establish recommendations for the diagnosis, prognosis, follow-up and treatment of the complications of MD1.
MATERIAL AND METHODS:
Consensus guide developed through a multidisciplinary approach with a systematic literature review. Neurologists, pulmonologists, cardiologists, endocrinologists, neuropaediatricians and geneticists have participated in the guide.
RECOMMENDATIONS:
The genetic diagnosis should quantify the number of CTG repetitions. MD1 patients need cardiac and respiratory lifetime follow-up. Before any surgery under general anaesthesia, a respiratory evaluation must be done. Dysphagia must be screened periodically. Genetic counselling must be offered to patients and relatives.
CONCLUSION:
MD1 is a multisystemic disease that requires specialised multidisciplinary follow-up.
Copyright © 2018 Sociedad Española de Neurología. Publicado por Elsevier España, S.L.U. All rights reserved.
KEYWORDS:
Clinical guideline; Complicaciones; Complications; Disfagia; Distrofia miotónica tipo 1; Dysphagia; Enfermedad de Steinert; Guía clínica; Myotonic dystrophy type 1; Recomendaciones; Recommendations; Steinert’s disease
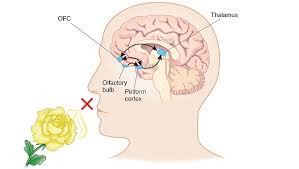
Smell Function (Odor recognition) may be affected by Myotonic Dystrophy
A new study that looked at MRI images showed that there were some affects to smell that could be seen on patients MRI. It is very possible that some people affected with DM may have reduced or impaired Smell (odor responses). The study is in Japanese so this is all the information that we can report here.
[Impairment of Odor Recognition in Myotonic Dystrophy Type 1].
Abstract
There is evidence that impaired human cognitive abilities are reflected in loss of olfactory abilities. Declining olfactory perception may be a biomarker for impairment of cognitive function and of impending illnesses in neurodegenerative disorders such as Parkinson’s disease (PD) and Alzheimer’s disease (AD). Previously, we reported that patients with myotonic dystrophy type 1 (DM 1) had lower sensitivity to emotional facial expressions as well as abnormal olfactory threshold or recognition level. In DM 1, pathological studies have reported neurofibrillary tangles in several temporal areas including the entorhinal cortex (ENT), hippocampus (HI), and the amygdala. We observed that patients with DM 1 showed signal abnormalities in the olfactory limbic areas on magnetic resonance imaging. Our findings underscore the need to pay careful attention to significant decreases in odor identification abilities caused by diverse forms of abnormal brain function, especially in the AMG, ENT and HI.