|
ACCOMMODATING PEOPLE WITH MUSCULAR DYSTROPHYAccording to the Muscular Dystrophy Association, the standard Myotonic Dystrophy is among the most common of Muscular Dystrophies with an incident of 1 in 7000 or 1 in 8000. Today, many individuals are living and working with MD, and employers are seeing more incidents of MD among their employees. This coupled with the requirements of the Americans with Disabilities Act (ADA) show why knowing about workplace accommodations for people with MD is important. When considering accommodations for people with MD, the accommodation process must be conducted on a case-by-case basis. Symptoms caused by MD vary so when determining effective accommodations, the person’s individual abilities and limitations should be considered and problematic job tasks must be identified. Therefore, the person with MD should be involved in the accommodation process. Not all people with MD will need accommodations to perform their jobs and many others may need only a few accommodations. For those who need accommodation, the following pages provide basic information about common limitations, symptoms, useful questions to consider, and accommodation possibilities. The following is only a sample of possibilities to consider; numerous other solutions and considerations may exist. Also included in this publication is a list of resources for additional information. This publication was written by Beth A. Loy, Ph.D., and Linda C. Batiste, MS, Human Factors Consultants with the Job Accommodation Network. If further information is needed, please call JAN at 1-800-526-7234 (V/TTY). 5/01 Muscular DystrophyWhat is muscular dystrophy?MD is the common name for several progressive hereditary diseases that cause muscles to weaken and degenerate. MD is caused by defects in genes for muscle proteins. Most of these proteins appear to play a role in supporting the structure of muscle fibers. The term “muscular dystrophy” refers to a group of genetic diseases marked by progressive weakness and degeneration of the skeletal, or voluntary, muscles, which control movement. The muscles of the heart and other involuntary muscles are also affected in some forms of MD, and a few forms involve other organs as well. There are forty different neuromuscular diseases; the major forms of muscular dystrophy are myotonic, Duchenne, Becker, limb-girdle, facioscapulohumeral, congenital, oculopharyngeal, distal, and Emery-Dreifuss. Some of these names are based on the locations of the affected muscles or the doctors who first described the diseases. For additional information on the major characteristics of the nine MDs, see Facts About Muscular Dystrophy (MD) from the Muscular Dystrophy Association at: <http://www.mdausa.org/publications/fa-md-9.html>. What causes muscular dystrophy?Flaws in muscle protein genes cause MD, which prevents the body from manufacturing essential substances in adequate amounts to maintain and fuel the muscles. All genes are inherited, half coming from a child’s mother, the other half from the father. Flaws in genes can be inherited along with the parents’ genes, although they can also occur for the first time in a child. Scientists use the term spontaneous mutation when this happens. The different forms of MD can be passed from parent to child by one of three inheritance patterns: 1) autosomal dominant (when only one parent passes on one defective gene), 2) autosomal recessive (when both parents pass on the same or similar gene defects), and 3) X-linked recessive (when gene defects on the X chromosome occur). What are the symptoms of muscular dystrophy?Individuals with MD usually exhibit contractures, a condition often associated with shortened muscles around the joints. Due to the abnormal and sometimes painful positioning of the joints, most individuals with MD have extreme fatigue and weakness as well as speech, mobility, and fine motor limitations. In addition, scoliosis, or curvature of the spine, is common. Who gets muscular dystrophy?MD is generally inherited but in some cases no family history of the disease may exist. MD can affect people of all ages. While some forms first become apparent in infancy or childhood, others may not appear until middle age or later. Duchenne and Becker are the most common types of MD and they occur in the male sex. X-linked recessive MD usually affect males since males do not have a “backup” copy of a normal gene on a second X chromosome. Females have two X chromosomes, so a normal gene on the female’s other X chromosome serves as a “backup” copy. A woman with an X-linked gene defect usually does not show any signs of an X-linked disease. However, she is a carrier of the disease and has a 50 percent chance of passing it to her sons. Her daughters each have a 50 percent chance of inheriting the gene and being carriers of the disorder. How is muscular dystrophy treated?Moderate exercise programs and physical therapy can minimize contractures, and certain exercises may prevent or delay scoliosis. Surgery can sometimes be helpful in relieving muscle shortening. In addition, respiratory care for some individuals with MD may also help. Medications known as corticosteroids have been found to slow muscle destruction in Duchenne MD. However, these potent anti-inflammatory drugs, used for many conditions besides MD, have serious side effects, such as weight gain, bone loss, cataracts, skin problems, high blood pressure, susceptibility to infection, and psychological difficulties. Researchers are testing new corticosteroids that may have fewer side effects. Researchers have developed accurate protein and DNA-based carrier detection tests for families affected by Duchenne and Becker MDs. Carrier testing for other types of MD is being developed. The prognosis of MD varies according to the type of MD and the progression of the disorder. Some cases may be mild and very slowly progressive, with normal lifespan, while other cases may have more marked progression of muscle weakness, functional disability and loss of ambulation. Life expectancy may depend on the degree of progression and late respiratory deficit. In Duchenne MD, death usually occurs in the late teens to early 20s. QUESTIONS TO CONSIDER WHEN DETERMINING ACCOMMODATIONSHow do these symptoms or limitations affect the person and the person’s job performance? What specific job tasks are problematic as a result of these symptoms and limitations? What accommodations are available to reduce or eliminate these problems? Are all possible resources being used to determine possible accommodations? Has the employee with MD been consulted regarding possible accommodations? Once accommodations are in place, would it be useful to meet with the person with MD to evaluate the effectiveness of the accommodations and to determine whether additional accommodations are needed? Do supervisory personnel and employees need training regarding MD, other disability areas, or the Americans with Disabilities Act? ACCOMMODATION CONSIDERATIONS FOR PEOPLE WITH MD(Note: People with MD will develop some of these limitations/symptoms, but seldom develop all of them. Limitations will vary among individuals. Also note that not all people who have MD will need accommodations to perform their jobs and many others may need only a few accommodations. The following is only a sample of the possibilities available. Numerous other accommodation solutions exist as well.) Activities of Daily Living:
Fatigue/Weakness:
Gross Motor:
Fine Motor Impairment:
Medical Treatment Allowances:
Speech Impairment:
Stress:
EXAMPLE ACCOMMODATIONS FOR PEOPLE WITH MDA student with MD was limited in her use of the computer. She was accommodated with the Magic Wand Keyboard, a miniature computer keyboard and mouse. The keyboard worked with the slightest touch of a wand and no force was needed to activate the keys. A staff employee with MD who operated a power chair with a joystick was having difficulty opening doors. The individual could not grasp door handles and was accommodated with automatic door openers. A physician with MD was having problems getting up from a seated position after consulting with patients. The individual was accommodated with a lift cushion for his chair. A counselor was having difficulty performing psychological evaluations due to cognitive limitations. Her manager agreed to provide written job instructions when possible, and make memory aids such as schedulers or organizers readily available. A service worker was having difficulty maintaining a full workday due to fatigue. The individual was accommodated with periodic rest breaks away from the workstation, a flexible work schedule, flexible use of leave time, parking close to the work-site, and periodic work from home. A technical writer with MD was having difficulty reaching her workstation. The individual was accommodated with a flat screen monitor, monitor arm, keyboard tray, footrest, headset, and strategically placed filing racks. A secretary with MD was restricted from typing information into her computer due to fine motor limitations. She was accommodated with speech recognition. A manager with MD was having difficulty with daily living needs. The individual was allowed to bring her service animal to work and provided an accessible restroom. A lawyer with MD was having difficulty climbing stairs. He was accommodated with a stair lift. An office worker with MD who distributed mail was having difficulty carrying mail to workstations. He was accommodated with a cart. A writer with MD had severe hand weakness and was limited in her ability to use the keyboard. The individual was accommodated with a miniature keyboard with light touch features. RESOURCES
|
Long Term Outlook Longevity
LONG TERM OUTLOOK LONGEVITY FOR PATIENTS
|
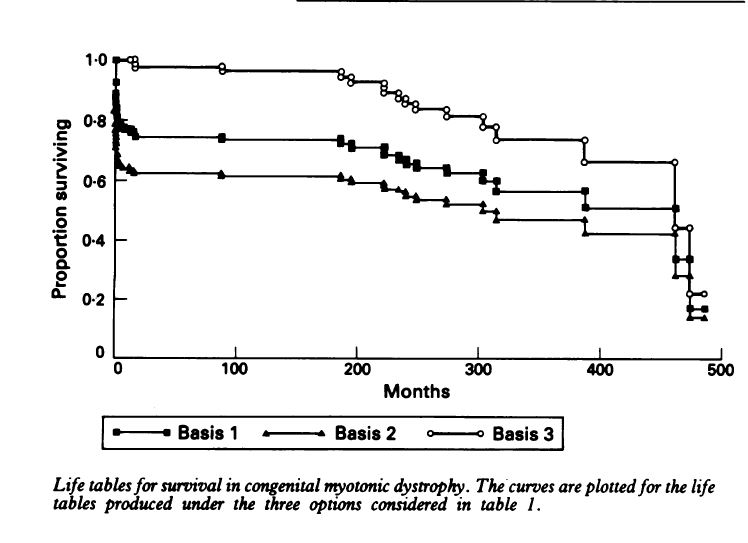
Myotonic Dystrophy is a slowly progressive neuromuscular Disease and the symptoms of the disease do gradually get worse over time. However, there are a number of ways to manage the symptoms of the disease and more and more management techniques are becoming available. In the very long-term outlook most people with DM will have shorter life expectancies, One study available at the end of this post gives an average of 59 years for females with DM1 and 60 Years for males For children with the Congenital form once beyond the early problems of respiratory distress the prognosis for life is relatively good. There will be improvement in early childhood and children will make steady progress in these early years. Motor function will improve, most children will walk and the marked hypotonia or floppiness will improve or disappear. For most children their performance will be limited by mental capacity and not by physical handicap. Reardon reports on the outcomes of 115 patients with confirmed diagnosis’s of CMD. The data suggests a 25% of death before 18 months of age and a 50% chance of survival into the mid-30’s. tables that follow are from Reardon. The Natural History of Congenital myotonic Dystrophy: Mortality and long term aspects. Archives of Disease in Childhood 1993; 68:177-181 Long-term Survival Probability
There was no instance of a congenitally affected patient having children. 48 patients reached the age of 20, 12 reached the age of 30. (of 115 patients). However, Reardon noted that “The degree of precision of estimated survival probabilities at the far end of the life table is recognized to be extremely poor” (or to put is more clearly he’s not sure how good the estimates are in the older years 30-40’s)During the study only 3 such individuals were studied and all died in this age band. Details of Current Employment among the 48 patients reaching 20 years of Age
Most of the patients with CMD will be unemployed. Chronic Medical Problems in surveying Patients n=71
Causes of Death n=44
However, during late childhood and early adolescence the “adult” features of myotonic dystrophy appear. Eye problems can be early on detected by the second decade and progressively muscles will be detected gradually weakening. No case of CMD has been found not to develop the adult version. O’Brien reports that of 46 patients with CMD 4 died outside the neonatal period at 4, 18, 19 and 22 years. Four more were considered seriously disabled with a poor prognosis. Problems seen in this group of 30 were gastrointestinal Problems in 8 (Constipation and abdominal pain) Talipes in 5. None in the group had any children and males showed marked testicular tubular failure indicating significant reproductive problems. Thus, long-term prognosis from a medical viewpoint is not bright. Reardon noted that because of gastrointestinal problems children with CMD will show a positive anal dilation test. This test is sometimes used to diagnosis sexual abuse. Parents with CMD should be aware of this information. This test should not be used for any conclusive evidence of sexual abuse with children with CMD. However, children with CMD are generally happy and cheerful individuals with a distinct personality and add a lot to family life as a whole. Medical outlook should be integrated with other family factors (see personal stories) New Study May 1999: A 10-year study of mortality in a cohort of patients with myotonic dystrophy. Mathieu J; Allard P; Potvin L; Pr´evost C; B´egin P Neuromuscular Clinic, Complexe Hospitalier de la Sagamie, Quebec University in Chicoutimi, Canada. Neurology, 52(8):1658-62 1999 May 12 Abstract OBJECTIVE: To determine the age and causes of death as well as the predictors of survival in patients with myotonic dystrophy (DM). METHODS: In a longitudinal study, a cohort of 367 patients with definite DM was followed for 10 years. RESULTS: During the 10-year period, 75 of the 367 DM patients (20%) died. The mean age at death (53.2 years, range 24 to 81) was similar for men and women. Among these 75 patients, 32 (43%) died of a respiratory problem, 15 (20%) of cardiovascular disease, 8 (11%) of a neoplasia, and 8 (11%) died suddenly. The ratio of observed to expected deaths was significantly increased to 56.6 (95% confidence interval [CI] 38.7 to 78.0) for respiratory diseases, 4.9 (95% CI 2.7 to 7.7) for cardiovascular diseases, and 2.5 (95% CI 1.1 to 4.6) for neoplasms. The mean age at death was 44.7 years for the childhood phenotype of DM, 47.8 years for the early-adult, 55.4 years for the adult, and 63.5 years for the mild phenotype (F = 4.8, p = 0.005). The age-adjusted risk of dying was 3.9 (95% CI 1.3 to 11.0) times greater for a patient with a distal weakness and 5.6 (95% CI 2.2 to 14.4) times greater for a patient with proximal weakness as compared with a person without limb weakness. CONCLUSIONS: Life expectancy is greatly reduced in DM patients, particularly in those with early onset of the disease and proximal muscular involvement. The high mortality reflects an increase in death rates from respiratory diseases, cardiovascular diseases, neoplasms, and sudden deaths presumably from cardiac arrhythmias. Here is another study abstract on Longevity: Age and causes of death in adult-onset myotonic dystrophy. Author de Die-Smulders CE; H¨oweler CJ; Thijs C; Mirandolle JF; Anten HB; Smeets HJ; Chandler KE; Geraedts JP Address Department of Clinical Genetics, Academic Hospital Maastricht, The Netherlands. christine.dedie@gen.unimaas.nl Source Brain, 121 ( Pt 8)():1557-63 1998 Aug Abstract Myotonic dystrophy is a relatively common type of muscular dystrophy, associated with a variety of systemic complications. Long term follow-up is difficult because of the slow progression. The objective of this study was to determine survival, age at death and causes of death in patients with the adult-onset type of myotonic dystrophy. A register of myotonic dystrophy patients was set up in Southern Limburg (the Netherlands), using data longitudinally collected over a 47-year period (1950-97). Survival for 180 patients (from the register) with adult-onset type myotonic dystrophy was established by the Kaplan-Meier method. The median survival was 60 years for males and 59 years for females. Survival of the patients was also estimated from the age of 15 years to the ages of 25, 45 and 65 years and compared with the expected survival of age- and sex-matched birth cohorts from the normal Dutch population. The observed survival to the ages of 25, 45 and 65 years was 99%, 88% and 18% compared with an expected survival of 99%, 95% and 78%, respectively. Thus, survival to the age of 65 in patients with adult-onset myotonic dystrophy is markedly reduced. A weak positive correlation between the CTG repeat length and younger age at death was found in the 13 patients studied (r = 0.50, P = 0.08). The cause of death could be determined in 70 of the 83 deceased patients. Pneumonia and cardiac arrhythmias were the most frequent primary causes of death, each occurring in approximately 30%, which was far more than expected for the general Dutch population. In addition, we assessed mobility in the years before death in a subgroup of 18 patients, as a reflection of the long-term physical handicap in myotonic dystrophy patients. Half of the patients studied were either partially or totally wheelchair-bound shortly before their death. |
More information is available on the lifespan on the congenital form of myotonic dystrophy from a study in 1993. Medical care has improved since then.
Pneumonia Lung Problems
LUNG PROBLEMS
Lung problems mostly occur during the advanced stage of the disease. Here too the weakness of the breathing muscles is not the main problem, because with MD in the worst case the breathing muscles are slightly affected. Chronic mechanical respiration, which is sometimes necessary with other muscular diseases, is seldom necessary with MD-patients.
The main problem with MD is the acute pneumonia. It is often caused by choking, though the patient is not always aware of it. Obviously he is choking on saliva when asleep, because swallowing is more difficult when lying down than being upright. Pneumonia mostly starts in the lower lobe of the lungs, but it can extend within a few days and become a serious and dangerous infection of a complete lung. This should be treated quickly with antibiotics and cough-medicine. Often admittance to hospital is necessary and sometimes patients require temporarily a mechanical ventilator at an Intensive Care. Sometimes the trachea must be sucked out with a bronchoscope, a flexible thin pipe, slid through the trachea by a lung-physician. Often this way of fighting pneumonia is successful. With some patients who frequently choke, within a few years several times pneumonia occurs. This can be prevented by sleeping at night in upright position, thus preventing choking.. In this case it is wise not to eat in the evening because from a filled stomach sometimes food is belched up, which can reach the lungs
Rehabilitation of Patients with Neuromuscular Disease
J Neuro Rehab 1997;11:69-80
©1997 Demos Vermande
Used with permission EDITOR NOTE THIS STUDY IS FROM 1997 about 22 years ago. Make sure to verify any information that you use from this study
From the Department of Rehabilitation Medicine
University of Washington, School of Medicine. Seattle, Washington.
Address correspondence and reprint requests to:
Gregory T. Carter. M.D., 500 S.E Washington, Chehalis, WA 98531
Supported by Research and Training Center Grant H133B30026 from the National Institute on Disability and Rehabilitation Research.
The goals of rehabilitation in patients with neuromuscular disease (NMD) are to maximize and prolong independent function and locomotion, inhibit physical deformity, and provide access to full integration into society. Management is best carried out by a multidisciplinary team consisting of physicians, nurses, therapists, social and vocational counselors, and psychologists, among others. Treatment is goal-oriented, using various modalities. Stretching, range of motion, timely surgical correction of spinal deformities and contractures, and bracing may improve or prolong ambulation as well as enhance functional use of the extremities. Moderate resistance (submaximal) weight lifting and aerobic exercise may improve strength and cardiovascular performance in slowly progressive NMDs. For patients with advanced restrictive lung disease, positive pressure ventilation may improve breathing and comfort. Cardiac complications in some NMDs can be severe and may require monitoring. Nutritional, psychological, and vocational considerations should also be part of the management of NMD. Major advances in biomedical and computer engineering continue to provide more functional equipment, allowing better strategies for improvement of quality of life. Key Words: Neuromuscular disease-Rehabilitation-Disability-Muscular dystrophy-Myopathy.
Introduction
Although currently incurable, neuromuscular diseases (NMDs) are not untreatable. The goals of rehabilitation in patients with NMDs are to maximize functional capacities, prolong or maintain independent function and locomotion, inhibit or prevent physical deformity, and provide access to full integration into society with good quality of life. The comprehensive management of the varied clinical problems associated with NMDs is an arduous task. For this reason, the multidisciplinary approach is much more effective and takes advantage of the expertise of many clinicians rather than placing the burden on one.
Management is best carried out by a team consisting of doctors, physical, occupational, and speech therapists, social workers, vocational counselors, and psychologists, among others. Treatment is goal-oriented, using various modalities. Impairment and disability should be thoroughly assessed based on the World Health Organization criteria as applied to neuromuscular diseases (Table 1). Impairment is evaluated by measurements of strength, range of motion, spine deformity, cardiac and pulmonary function, and intellectual capacity. Disability is evaluated by measures of mobility and upper extremity function, cardiopulmonary adaptations, cardiac and pulmonary complications, and psychosocial adjustment.
| Table 1. Disability in neuromuscular diseases. World Health Organization (WHO) definitions of impairment, disability, and disadvantage, based on organ systems |
|||
| Organ | Impairment (usually progressive) |
Disability | Disadvantage (handicap) |
| Skeletal muscle | Strength and endurance | Motor performance Mobility |
Quality of life Educational opportunities |
| Upper extremity function | Employment opportunities | ||
| Fatigue | Dependency and disadvantage | ||
| Bone & joint | Joint Contractures Spine Deformity |
Function Pain and deformity |
|
| Lungs | Pulmonary function | Restrictive Lung Disease (RLD) Fatigue |
|
| Heart | Cardiomyopathy Conduction defects |
Cardiopulmonary adaptations Fatigue |
|
| CNS | Intellectual capacity | learning ability Psychosocial adjustment |
|
|
|
|||
Ideally, due to the significant mobility problems associated with most NMDs, all key clinic personnel should be available at each visit. Tertiary care medical centers in larger urban areas can usually provide this type of service. This may be an independent clinic or a clinic funded by one or more of the consumer driven organizations sponsoring research and clinical care for people with NMDs, including the Muscular Dystrophy Association (MDA). Governmental agencies that support research in NMDs include the National Institute on Disability and Rehabilitation Research (NIDRR), which is a division of the Department of Education, as well as the National Institutes of Health (NIH).
An accurate confirmation of the diagnosis is critical and involves thorough clinical evaluation electrodiagnostic studies, and often a muscle or nerve biopsy. For many of the diseases, DNA analysis of leukocytes or other cellular components obtained through a blood draw is commercially available and contributes greatly to the accuracy of the diagnosis.
Once the diagnosis is confirmed, the patient and family should be thoroughly educated about the expected outcome and what problems may be encountered. The physician should then assess the goals of patient and family and orchestrate a palliative and rehabilitative program that matches those goals. Enrollment in an experimental protocol should be encouraged and facilitated; this not only furthers science but also provides some hope for the patient.
Overview of the Major Neuromuscular Diseases
Duchenne and Becker Muscular Dystrophy
Both Duchenne muscular dystrophy (DMD) and Becker muscular dystrophy (BMD are inherited X-linked recessive diseases affecting primarily skeletal muscle and myocardium. Dystrophin, a large cytoskeletal protein in the subsarcolemmal lattice, which stabilizes the plasma membrane during muscle contractions, is absent in DMD and of abnormal molecular weight and/or reduced amounts in BMD (1). This results in an unstable muscle cell membrane and impaired intracellular homeostasis, causing muscle fibers to continually deteriorate and regenerate until the capacity for repair is no longer sufficient and the muscle fibers undergo irreversible degradation with replacement by fat and connective tissue (1,2). The abnormal gene for DMD and BMD is on the short arm of the X chromosome at position Xp21 (1,2).
DMD is the most common neuromuscular disease of childhood, with prevalence rates ranging from 19 to 95 per million and an estimated overall prevalence of 63 per million (3). This progressive myopathy is universally fatal, with death usually occurring from respiratory or cardiac complications. BMD has a lower incidence than DMD, with prevalence rates for BMD ranging from 12 to 27 per million and a recently estimated overall prevalence of 24 per million (2,3). BMD shows a similar pattern of muscle weakness to DMD but with later onset and much slower rate of progression (4).
Myotonic Muscular Dystrophy
Myotonic muscular dystrophy (MMD), an autosomal dominant multisystem hereditary muscular dystrophy with an incidence of 1 in 8,000, is characterized clinically by progressive, predominantly distal muscle weakness and myotonia. Associated findings include frontal baldness, gonadal atrophy, cataracts, and cardiac dysrhythmias (3,5). Two types are recognized: noncongenital (NC-MMD) and congenital (C-MMD). In MMD a DNA sequence within the gene on chromosome 19q 13.3, is repeated many times, leading to an enlarged, unstable area of the chromosome. Called a triplet repeat mutation, the flawed gene grows by sudden leaps when transmitted from generation to generation, causing the disease to occur at a younger age and in a more severe form (5,6). C-MMD patients have been shown to have substantially more repeats than those found in NC-MMD patients. The repeated DNA sequences known as CTG (trinucleotide cytosine, thymine guanidine) are linked to the production of a protein (myotonin-protein kinase) that has important functions in smooth and skeletal muscle, eye, hair, and brain, and decreased levels of messenger RNA and protein expression (5,6).
Facioscapulohumeral Dystrophy
Facioscapulohumeral dystrophy (FSHD) is a slowly progressive myopathy with autosomal dominant inheritance and prominent involvement of facial musculature. Prevalence is difficult to ascertain due to undiagnosed mild cases but has been estimated at 10-20 per million (3). The abnormal gene is known to be near the end of chromosome 4, although exact DNA testing for diagnostic purposes is not yet available except possibly in cases in which a detailed genetic analysis of a family exists (7). FSHD can be quite heterogeneous in its clinical presentation and course, which leads to questions regarding genetic homogeneity (7). Sporadic forms may be referred to as FSH syndrome (FSHS).
Limb-Girdle Syndrome
Limb-girdle syndrome (LGS), a heterogeneous group of myopathies that share some clinical features, has been classified into five types: autosomal recessive muscular dystrophy of childhood (ARMDC), autosomal dominant late-onset (ADLO), pelvifemoral (PF:), scapulohumeral (SH), and myopathy limited to quadriceps (8,9). In any of these, expression may be in either sex, with primary involvement of the shoulder and/or pelvic girdle muscles and a variable rate of progression. LGS is no longer considered a distinct nosological entity and is thought to be a wide variety of muscle disorders predominantly affecting the limb girdle muscles. Genetic studies have identified one form linked to chromosome 15q, another form to chromosome 2p, and two more severe forms to 13ql2 and 17ql2-q21 (8,9). Defects at the chromosome 13q locus and 17q locus result in a specific abnormality in the transmembrane dystrophin associated glycoprotein. and patients with this defect may clinically resemble severe BMD despite the normal expression of dystrophin (10).
Late-onset limb girdle muscular dystrophy with autosomal dominant inheritance has been linked to chromosome 5 but may also be present in childhood and referred to as Bethlem myopathy (8). Pelvifemoral LGS is so heterogeneous that its existence as a discrete disorder remains controversial. Due to substantial heterogeneity, prevalence rates for these syndromes have not been reported.
The Charcot-Marie-Tooth Syndromes
Also known as hereditary motor and sensory neuropathy (HMSN), Charcot-Marie-Tooth (CMT) syndromes are among the most common hereditary neuromuscular diseases, with prevalence ranging from 14 to 282 per million (3 ). There are at least eight forms of HMSN. Types I, II, and III represent the Charcot-Marie-Tooth syndrome. HMSN I (hypertrophic nerve), the most common HMSN, is characterized by markedly reduced conduction velocities in peripheral motor and sensory nerves. HMSN II (neuronal form) exhibits predominant axonal loss, while conduction velocities remain relatively normal. HMSN II is usually a less severe disease than HMSN I, although clinically it may not be easily distinguished from type I. HMSN III is a congenital hypomyelinating neuropathy, with symptoms beginning in infancy (11).
There are several notable subtypes. HMSN type 1a has a DNA duplication at the 17p11.2 locus for the peripheral myelin protein 22 (PMP22) gene. Point mutations in the PMP22 gene have recently been identified in some HMSN 1a patients who did not exhibit the duplication (11,12). The defect in HMSN type 1b is located on chromosome 1 with another myelin protein defect (12,13). Five families have been reported with autosomal dominant HMSN 1b mapping to chromosome 1q21-23 (13). Additionally, autosomal dominant HMSN 1c, which does not map to chromosome 1 or 17, has been reported (13,14).
Spinal Muscular Atrophy
There are at least three forms of spinal muscular atrophy (SMA), all of which involve selective destruction of anterior horn cells (15). These disorders are inherited, autosomal recessive traits. SMA I, also known as Werdnig-Hoffman disease (WHD) or acute, infantile-onset SMA, is a severe disorder that results in death before the age of two years. SMA II, also referred to as early-onset, intermediate SMA, or chronic Werdnig-Hoffman disease, is less severe, with signs and symptoms becoming apparent in the first 6 to 18 months of life. SMA III, also known as Kugelberg-Welander disease (KWD), is a chronic, later onset disorder, associated with significantly less morbidity. Signs and symptoms of SMA III become apparent between ages 5 and 15 years (15). Genetic studies indicate that all types of SMA may be caused by different defects occurring in genes located on the long arm of chromosome 5q13.1, including the survival motor neuron gene and the neuronal apoptosis inhibiting protein (NAIP) gene (15,16).
Prevalence rates for types II and III range from as high as 40 per million among children to around 12 per million in the general population (3).
Amyotrophic Lateral Sclerosis
Amyotrophic lateral sclerosis (ALS), like SMA, is a motor neuron disorder (MND). ALS rapidly destroys upper and lower motor neurons, causing spasticity and diffuse muscular atrophy and weakness. Most ALS cases are acquired and occur sporadically. Approximately 1% of ALS cases result from a familial trait due to a specific gene defect on chromosome 21q12.1, leading to an abnormality in the production of the antioxidant enzyme Cu/Zn superoxide dismutase (SOD1) (17). Current popular theories regarding the etiology of sporadic ALS include excessive glutamate activity in the brain and spinal cord due to defective cellular transport mechanisms and/or possible deficiencies in various neurotrophic growth factors (17,18).
ALS most commonly strikes people between 40 and 60 years of age, with a mean age of onset of 58 yrs. (19,20,21). The overall prevalence rate in the worldwide population is between 5 and 7 per 100,000. making it one of the most common neuromuscular diseases worldwide (3,20.21) Overall, he median 50% survival rate is 2.5 yrs. (19,20,21).
Spectrum of Clinical Problems and Treatment Paradigms
Weakness and the Role of Exercise
Skeletal muscle weakness is the ultimate cause of the majority of clinical problems in NMDs. There have been several well-controlled studies looking at the effect of exercise as a means to gain strength in NMDs (22,23,24,25). In slowly progressive NMDs, a 12-week moderate resistance (30% of maximum isometric force) exercise program resulted in strength gains ranging from 4% to 20% without any notable deleterious effects (23). In the same population, a 12-week high resistance (training at the maximum weight a subject could lift 12 times) exercise program showed no further added beneficial effect compared to the moderate resistance program, and there was evidence of overwork weakness in some subjects (24). In a comparative study, CMT patients appeared to benefit significantly from a strengthening program whereas MMD patients showed neither beneficial nor detrimental effects (25). It is likely that the most effective exercise regimens for neuropathies and myopathies will be different, although further studies are needed to confirm this. Due to the active, ongoing muscle degeneration in the rapidly progressive disorders DMD and ALS, the risk for overwork weakness is great and exercise should be prescribed cautiously with a common sense approach.
Dystrophin deficient muscle is very susceptible to exercise-induced muscle injury (26). Using voluntary running protocols, we studied the effect of exercise on skeletal muscle from both normal and mdx mice, a genetically homologous murine model of DMD (27,28,29). In contrast to normal mice, mdx mice show considerable avoidance behavior for exercise. which may be an intuitive survival strategy. After exercise on a mouse wheel ad libitum, the extensor digitorum longus (EDL) and soleus muscles of adult mdx mice became significantly weaker and showed histochemical signs of further damage (figure 1) compared to control mdx muscles (27).
![[Figure 1]](http://www.myotonicdystrophy.org/carter-cell.jpg) Figure 1. Photomicrograph of quadriceps muscle from adult mdx mouse 3 days following running exercise and subsequent tail vein injection of 10,000 molecular weight fluorescent dextran. There is significant intracellular staining in several fibers (center), indicating membrane damage. Central nuclei, present in regenerating fibers, are shown by focal intense areas of fluorescence. Figure 1. Photomicrograph of quadriceps muscle from adult mdx mouse 3 days following running exercise and subsequent tail vein injection of 10,000 molecular weight fluorescent dextran. There is significant intracellular staining in several fibers (center), indicating membrane damage. Central nuclei, present in regenerating fibers, are shown by focal intense areas of fluorescence. |
All patients with NMDs should be advised not to exercise to exhaustion due to the risk of muscle damage and dysfunction. Patients participating in an exercise program should be cautioned of the warning signs of overwork weakness, which include feeling weaker rather than stronger within 30 minutes post exercise or excessive muscle soreness 24-48 hours following exercise. Other warning signs include severe muscle cramping, heaviness in the extremities, and prolonged shortness of breath.
Nonetheless, early intervention with gentle, low impact aerobic exercise like walking, swimming, and stationary bicycling will improve cardiovascular performance and increase muscle efficiency and thus help fight fatigue. Fatigue in NMDs is multifactorial and is due in part to impaired muscular activation, generalized deconditioning, and diminished cardiopulmonary performance from immobility (30,31). Aerobic exercise not only improves physical functioning but also is beneficial in fighting depression, maintaining ideal body weight, and improving pain tolerance.
Exercise may induce muscle cramping in NMD, presumably due to sarcolemmal instability (27,29). Exercise may also cause symptomatic muscle spasms related to spasticity in ALS (30). Baclofen acts to facilitate motor neuron inhibition at the spinal levels and is a good initial agent to use. The benzodiazepines and other centrally acting muscle relaxants may induce somnolence and respiratory suppression. Dantrolene, by impairing excitation-contraction coupling, is effective at reducing muscle tone but also causes generalized muscle weakness and should not be used. Membrane stabilizing agents like tocainamide, mexilitine, and phenytoin may also be helpful, particularly in the myotonic disorders. Amitriptyline, although reportedly effective in relieving myotonia in human MMD, does not have any effect on normal or myotonic mouse skeletal muscle (32). Slow (30 second sustained), static muscle stretching may also be helpful.
Although still considered the clinical standard, manual muscle testing for assessment of strength in NMD has been shown to be unreliable (33,34,35,36). A hand-held myometer or MicroFET® type of strength measuring device is far more reproducible and as easy to use in a clinical setting (33,34). Quantitative methods, including isokinetic measurements, are reliable yet require more sophisticated equipment and usually cannot be done in the clinic (33,36). NMDs may present with fairly asymmetrical patterns of weakness. The notable exception is CMT, where marked asymmetry in strength should raise the possibility of a concomitant focal entrapment neuropathy or other diagnosis (37).
Limb Contractures and Scoliosis: The Role of Stretching, Bracing, and Surgery
Although there is some variability in reported incidence, significant joint contractures and scoliosis develop in a large percentage of DMD and SMA II patients (2,15,18). Joint contracture and scoliosis may occur but are not usually major clinical problems in the other NMDs (18). All NMD patients should have an examination of the spine and major joints at each clinic visit.
![[Figure 2]](http://www.myotonicdystrophy.org/carter-shoulder.jpg) Figure 2. Profound shoulder girdle muscular atrophy and subluxation in an adult with facioscapulohumeral dystrophy. Figure 2. Profound shoulder girdle muscular atrophy and subluxation in an adult with facioscapulohumeral dystrophy. |
The occurrence of contractures appears to be directly related to prolonged static positioning of the limb, and these contractures often develop soon after wheel-chair reliance (38,39). Several studies have shown that wheelchair reliance and lack of lower extremity weight bearing contribute to the rapid acceleration of contractures (2,38). Upper extremity contractures may occur in ambulatory patients with focal, proximal atrophy, particularly at the shoulder girdle (Figure 2). This may be worsened by subluxation. Slings may be helpful to support the joint but do not prevent contracture formation or subluxation. Gentle static stretching and splinting may slow the progression contractures but this has not been well studied. Although orthopedic contracture release allows a patient to be braced and may prolong ambulation, it appears that weakness is the major factor that inhibits ambulation, not joint contracture per se. In a randommed tral, Manzur and colleagues showed no benefit to early surgical treatment of contractures in DMD (40). Studies indicate that weakness is the major cause of loss of ambulation in DMD, not contracture formation (2,38). Wheelchair reliance and scoliosis were both found to be age-related phenomena but not necessarily linked causally. Several investigators note no relationship between age of wheelchair reliance and subsequent progression of scoliosis (2,38). Lord and colleagues reported a 3.5 year difference between wheelchair dependency and scoliosis in DMD (41). The causal relationship between loss of ambulatory status and scoliosis is doubtful given the substantial time interval between the two variables in most subjects. Further, a significant percentage of DMD and some SMA patients develop scoliosis before they become wheelchair-dependent (2,15). It is more likely that both wheelchair reliance and scoliosis are related to other factors (e.g., age, adolescent growth spurt, increasing weakness of trunk musculature, or other unidentified factors) and thus represent coincidental signs of disease progression.
DMD patients develop scoliosis most commonly between the ages of 13 and 15 years, corresponding closely with the adolescent growth spurt (2). While there is evidence on computerized tomography for asymmetric involvement of the erector spinae in DMD, it is extremely difficult to clinically detect asymmetric paraspinous muscle strength (42). Studies of trunk flexor or extensor strength in NMD patients with or without scoliosis showed no significant difference in MMT, although this method is not capable of measuring asymmetric strength (2,42,43). Several studies confirm that primary or single thoracolumbar curves are more common than lumbar curves (44,45,46,47). Spinal bracing has not been shown to be effective in preventing progression of scoliosis in NMD (48,49). Patients with DMD and SMA should be followed closely with serial radiographs and have appropriate and timely spinal instrumentation and fusion, which should be done before the primary curve becomes greater than 25° and vital capacity (VC) has not gone below 50% of predicted (50,51,52). Although progressive neuromuscular scoliosis does create significant positioning and musculoskeletal problems, it has not been shown to directly affect pulmonary function (53,54,55). Complications increase substantially if the patient already has compromised breathing. If surgery is done in curves that have progressed much beyond 40°, the likelihood of successful correction and fusion is diminished (2,52,53,54,55).
Bracing to Prolong or Maintain Ambulation
The purpose of bracing at any level is to improve function and stability. The use of long leg bracing prolongs ambulation time and has been advocated in DMD partly on the presumption that this will delay scoliosis (2,48,51,52). Several studies have suggested that the ability to continue ambulation with long leg braces for several years may also be indicative of a slower disease progression rate with relatively less weakness of trunk musculature present at a given age (2,30). Any association between prolonged ambulation with long leg bracing and delayed or decreased scoliosis in DMD may not be directly causal. Due to weakness in both proximal (hip and knee extension) and distal (ankle plantar and dorsiflexion) muscle groups in DMD, a long leg brace or knee-ankle foot orthosis (KAFO) is generally needed (2,18,30).
Most CMT patients require short leg braces or ankle-foot orthoses (AFOs). These should be custom-made of a lightweight polymer such as polypropylene or carbon filer resin and should fit intimately to provide good stability and prevent pressure sores. The older, traditional double metal upright AFOs built-in to the shoe may be too heavy and can actually hinder ambulation. If there is severe distal muscle weakness and ankle instability, the braces should be high profile, coming around the ankle in front of the malleoli. A pes cavus foot with a hammertoe deformity can be accommodated with a built-up arch and metatarsal bar. Patients with CMT are at high risk for neuropathic arthritis (Charcot joint) and skin ulcers. Their skin and weight-bearing joints should be closely examined at every clinic visit.
Patients with other NMDs may also benefit from bracing, depending on the distribution of weakness, gait problems, and joint instability created by the weakness. In making the decision to brace, the clinician should also consider the added weight of the brace and whether or not the patient is willing to use a brace. If there are questions about whether or not a brace would help, the patient should be tried in an off-the-shelf model first, along with careful assessment by the clinician. Most patients require a short course of physical therapy after the braces are made to help them use the braces effectively.
Restrictive Lung Disease
Restrictive lung disease (RLD) is common in most NMDs due to weakness of the diaphragm, chest wall, and abdominal musculature (2,15,18,56). RLD can become quite severe in DMD, SMA, MMD, and ALS. Patients should be educated early in the disease process so informed decisions can be made farther down the line (18,56,57,58,59). Routine pulmonary function tests (PFTs), including forced vital capacity (FVC) and maximal inspiratory and expiratory pressures (MIP/MEP), should be monitored closely. The MIP reflects diaphragmatic strength and the ability to ventilate. The MEP reflects chest wall and abdominal muscle strength and the ability to cough and clear secretions.
Ultimately, most patients with severe RLD develop hypoventilation, which leads to elevated CO2 levels (18,56,58,59,60). Measuring only O2 saturation levels with pulse oximetry may be inadequate. End tidal CO2 levels or arterial blood gases (ABGs) should be measured periodically, depending on the clinical condition of the patient. A thorough review of systems will help define any problems. Patients who are hypoventilating often become hypercapnic at night and complain of a morning headache, restlessness or nightmares, and poor quality sleep. This may cause daytime somnolence. Insufficient respiration with hypoxia may occur later, especially if the lung is damaged by chronic aspiration.
Options for noninvasive interventions include a chest cuirass or porta-lung, which mechanically inflates the lung by creating negative chest wall pressure, similar to the iron lungs commonly used during the polio out-breaks. Although effective, these devices are cumbersome and may be poorly tolerated (18,56,57,60). Intermittent positive pressure ventilation (IPPV) by mouth avoids the need for tracheostomy and maintains reasonable quality of life. Bimodal positive airway pressure (BiPAP) is the best initial form. This can be done in the home and should be considered the preferred modality of assisted ventilation in NMDs (57). It generally takes some work with a respiratory therapist to get a good face or lip seal on the mask or nasal/oral orthotic interface. Patients may benefit initially from using assisted ventilation mainly at night. If better airway access becomes absolutely necessary and the informed patient desires more aggressive care, a tracheostomy may be done.
There have been numerous case reports of respiratory failure in people with CMT, the etiology of which has remained elusive. Electrodiagnostic and pathologic studies on the phrenic nerve in CMT confirm that it is involved in the disease (61,62). In one study phrenic nerve latency was abnormally prolonged in 96% of the CMT subjects, but significant PFT abnormalities and clinical symptoms were uncommon and did not correlate with phrenic nerve latencies (61). Although phrenic nerve latencies are markedly prolonged in CMT, they are not useful in predicting respiratory dysfunction.
Bulbar palsy and dysphagia may occur in ALS and some rare forms of SMA (15,18,30). Early signs of dysphagia include changes in voice patterns (voice becoming hoarse) and persistent coughing after swallowing liquids, which can indicate micro-aspiration. A speech therapist should be consulted early on for clinical swallowing evaluations, and recommendations on dietary modification. Thickening liquids and preparing food that forms a bolus easily will help. A modified barium swallow (MBS) is helpful in accurately determining the presence of aspiration as well as in defining which food textures the patient can safely swallow (63).
A percutaneous endoscopic gastrostomy (PEG) cube may be needed for nutrition. If necessary, a laryngeal diversion (laryngotracheal separation) procedure has several distinct advantages over tracheostomy (64). In this procedure, the trachea is surgically separated and a cutaneous tracheostoma is formed with the distal segment. The proximal trachea is either oversewn or sewn side-on-end into the esophagus. This procedure completely eliminates the possibility of aspiration and requires much less deep suctioning than tracheostomy. The tracheostoma does not require any hardware (such as a tracheostomy tube), and the patient may still take some food for pleasure without risk of aspirating. The primary disadvantage is complete loss of phonation since air no longer flows through the vocal cords, so it is only recommended when there is severe dysarthria accompanying the dysphagia and the patient’s speech is unintelligible. While preserving the ability to phonate, a tracheostomy actually increases aspiration risk, requires significantly more care, and provides no better airway access (64).
Cardiac Complications
Significant cardiac involvement occurs in DMD, BMD, MMD, and some cases of the ARMDC subtype of LGS (2,4,5,8). There is a high (60%-80%) occurrence of cardiac involvement in DMD and BMD subjects across all ages (2,4,65,66). Electrocardiogram (ECG) and echocardiographic abnormalities in DMD and BMD are not generally associated with a high incidence of early clinical heart disease in the pre-adolescent years. Dystrophin has been localized to the membrane surface of cardiac Purkinje fibers, and defective dystrophin expression likely contributes to the cardiac conduction disturbances seen in DMD and BMD (67).
In spite of significant ECG and echocardiographic abnormalities, only about 30% of DMD patients have clinical cardiovascular complications (2,65.66). Myocardial impairment remains clinically silent until late in the course of the disease, possibly due to the absence of exertional dyspnea secondary to lack of physical activity. Pulmonary hypertension has also been implicated in the cardiorespiratory insufficiency of DMD. Death has been attributed to congestive heart failure in as many as 40% of patients with DMD by some investigators (65.66).
The pattern of occasional life-threatening cardiac involvement in BMD may be more rapid than the progression of skeletal myopathy (4,18,65). The cardiac compromise may be disproportionately severe relative to the degree of restrictive lung disease in some BMD subjects. The evidence for significant myocardial involvement in BMD is sufficient to warrant screening of all these pa-tients at regular intervals using ECG and echocardiography. Close follow-up is needed for those with myocardial involvement. The slowly progressive nature of this dystrophic myopathy, which is compatible with many years of functional mobility and longevity, makes these patients suitable candidates for cardiac transplantation if end-stage cardiac failure occurs. Successful cardiac transplantation has been increasingly reported in BMD subjects with cardiac failure who are otherwise still quite physically functional (4).
Electrocardiographic abnormalities have been extensively reported in myotonic muscular dystrophy (5). Approximately 33% of NC-MMD subjects had first degree AV block, 17% had left axis deviation, and 5% had left bundle branch block and, rarely, complete heart block requiring pacemaker placement. Although few subjects show clinical signs and symptoms of cardiomyopathy, sudden death does occur in relatively young NC-MMD subjects and bundle of His conduction delays have been reported (5). Any MMD subject with dyspnea, chest pain, or cardiac symptoms should receive thorough cardiac evaluation including bundle of His studies.
Cardiac involvement has only recently been recognized in LGS patients (8). As in BMD, the cardiac manifestations in LGS may predate the clinical evidence of skeletal myopathy (8,68). Approximately 22% of ARMDC patients demonstrate left ventricular hypertrophy on ECG. Left ventricular hypokinesis with a compensatory resting tachycardia, as seen in DMD, is not a common finding in ARMDC and intervention is rarely warranted (2,8,68).
Nutritional Management
In more severe NMDs there is an increasing tendency toward obesity around or after loss of independent ambulation. Obesity is common in NMDs, particularly DMD where a 54% prevalence has been reported (2,69). In prior studies obesity was unrelated to strength decline. age of wheelchair reliance, functional grade status, timed motor performance, pulmonary function, likelihood of ECG abnormalities, or age at death (2). Weight control has its primary rationale in ease of care, particularly ease of transfers and skin care.
Malnutrition may be a significant problem in advanced stages of DMD, ALS, and SMA. Nutritional compromise becomes particularly problematic if there is severe RLD with increased work of breathing, which may also influence caloric intake. Self-feeding often becomes impossible during this period. DMD patients may develop signs and symptoms of upper gastrointestinal dysfunction, making feeding more difficult (70). Nutritional counseling with energy intake and energy allowance estimations should be done for all NMD patients with severe RLD (FVC < 50% predicted). Placement of a PEG tube may facilitate nutrition even if the patient can swallow because it allows for rapid and easy delivery of large amounts of calories and fluids. Patients should be reassured that they will still be able to eat food orally for enjoyment.
Psychosocial and Vocational Considerations
The majority of people with NMDs show intellectual levels within the normal range. However, approximately 30-50% of DMD and MMD patients will manifest mild clinical impairment (2,4,5,7, 8,11,15,71). Nonetheless, a large percentage of NMD patients exhibit elevated scores for hypochondriasis, depression, and hysteria on the Minnesota Multiphasic Personality Inventory (MMPI) test (2,4,5,7,8,11,15). In one study these indicators of emotional pathology were associated with unemployment, whereas loss of ambulation and arm function were not. Overall, employment rates are substantially lower for people with NMDs than for the able-bodied (18). Despite this, a higher level of education correlated with higher employment rate and improved self-esteem in this group (18). This implies that altered personality profiles in people with NMDs substantially impact social integration and employment rates, and, indeed, may be as important as physical abilities with respect to this. Education appears to be very important with respect to employability and self-esteem and should be emphasized in people with NMDs.
Patients should be referred to a support group. Support groups are often a great resource, not only for psychological support but also for problem solving and recycling of equipment such as hospital beds.
Reactive clinical depression may occur in advanced DMD and ALS. Good family, social, and religious support systems are helpful. Antidepressant medication may provide assistance with mood elevation, appetite stimulation, and sleep. Antidepressants with significant anticholinergic activity may help dry oral secretions in ALS and minimize drooling. Referral to a psychiatrist or clinical psychologist may be required. Depression in the spouse or significant other, family, or friends should not be overlooked and group/family counseling may be helpful.
Equipment
There are a number of additional useful equipment items that can substantially improve quality of life, including a hand-held shower, bath tub bench, grab bars, raised toilet seat, hospital bed, commode chair, ADL aids (sock aid, grabbers, etc.), and wheelchair ramp. Wheelchairs should be properly fitted (generally by an occupational therapist) and must have adequate lumbar support and good cushioning to avoid pressure ulcers. Simply giving the patient a prescription for a wheelchair often results in the patient’s getting a standard manual chair that does not fit properly. Although expensive, an electric wheelchair can be justified on the basis that it will help prolong independent mobility and thus markedly improve quality of life.
A good pressure-relieving mattress (air or dense foam) should be used on the bed at home, along with foam wedges to facilitate proper positioning. This helps prevent pressure ulcers and contractures. Wheeled walkers or quad (four point) canes may also help, depending on the pattern of weakness. Frequently severe weakness in the neck flexors and extensors cause a “floppy head” associated with severe neck pain and tightness. This may be helped by a hard cervical (Philadelphia type) or Freeman collar. Communicative aids including an alphabet, word board, or speech synthesizer help with dysarthria in ALS. An occupational therapist can help define which, if any, of these devices will be useful to the patient.
Areas of Future Research
Major advances in the understanding of the molecular basis of many NMDs has greatly enhanced diagnostic accuracy and may provide the basis for therapeutic intervention. There are many promising strategies developing for gene insertion and DNA repair, which are already impacting prenatal diagnostic capabilities and may someday lead to the ability to reverse or even cure some NMDs (72).
There have also been major pharmacologic advances. Riluzole is a neuroprotective agent that appears to inhibit glutaminergic neurotransmission in the spinal cord (73). It is the first agent approved by the Food and Drug Administration (FDA) for use in ALS. Although it has only a modest beneficial effective at improving life expectancy in ALS, it is nonetheless a major advance-ment (73). Various neurotrophic growth factors also appear promising, some of which are now being tried via intraventricular delivery systems (74).
Although not FDA-approved for this use, prednisone, at 1 mg/kg/day, given to DMD boys between ages 4 to 8 years old, has been shown to prolong time of ambulation and should at least be considered for use in this disease, although major side effects include weight gain, osteoporosis, and mood lability (75). Important advances in the use of reliable functional assessment tools have made it easier to judge the effectiveness of experimental interventions. The Timed Motor Performance assessment (Table 2) developed by Brooke and co-workers is one good example of a simple measurement scale that can be used at routine clinic visits (76).
Major advances have been made in the fields of biomedical engineering and computer science, providing patients with NMDs refined, more functional equipment and allowing better strategies for improvement of quality of life. As progress continues to change our management of NMD, it also changes the expectations of patients. Many patients with severe NMD may now live through child-bearing years, possibly bearing children, and expecting to enjoy a high quality of life (77). A comprehensive approach to management of the NMD patient can often fulfill these expectations.
Table 2. Timed motor performance tasks. Time to perform is measured in seconds with a stopwatch. Failure is considered inability to complete the task by a time limit of 120 seconds.
![]()
- Standing from lying supine.
- Climbing 4 standard stairs (beginning and ending standing with arms at sides).
- Running or walking 30 feet (as fast as is compatible with safety).
- Standing from sitting on chair (chair height should allow feet to touch floor).
- Propelling a wheelchair 30 feet.
- Putting on a T-shirt (sitting in chair – see instructions).
- Cutting a 3″ x 3″ premarked square from a piece of paper with safety scissors (lines do not need to be followed precisely).
Glossary of Acronyms
| ADLO: | Autosomal dominant late onset muscular dystrophy |
| AFO: | Ankle-foot orthosis |
| ALS: | Amyotrophic lateral sclerosis |
| ARMDC: | Autosomal recessive muscular dystrophy of childhood |
| BMD: | Becker muscular dystrophy |
| C-MMD: | Congenital myotonic muscular dystrophy |
| CMT: | Charcot-Marie-Tooth syndromes |
| DMD: | Duchenne muscular dystrophy |
| FSHD: | Facioscapulohumeral dystrophy |
| HMSN: | Hereditary motor and sensory neuropathy |
| HMSN 1: | Hereditary motor and sensory neuropathy (hypertrophic nerve) |
| HMSN 11: | Hereditary motor and sensory neuropathy (neural form) |
| HMSN 111: | Hereditary motor and sensory neuropathy (congenital hypomyelinating neuropathy) |
| KAFO: | Knee-ankle foot orthosis |
| LGS: | Limb girdle syndrome |
| MDA: | Muscular Dystrophy Association |
| MMD: | Myotonic muscular dystrophy |
| MMT: | Manual muscle testing |
| NCMMD: | Noncongenital myotonic muscular dystrophy |
| NMD: | Neuromuscular disease |
| PF: | Pelvifemoral muscular dystrophy |
| RLD: | Restrictive lung disease |
| SMA: | Spinal muscular atrophy |
| SMA II: | Early onset or chronic Werdnig-Hoffman disease |
| SMA III: | Kugelberg-Welander disease |
| VC: | Vital capacity |
References
- Hoffman EP, Brown RH Jr, Kunkel LM. Dystrophin: the protein product of the Duchenne muscular dystrophy locus. Cell 1987; 51:919-28.
- McDonald CM. Abresch RT, Carter GT, Fowler WM, Johnson ER, Kilmer DD. Profiles of neuromuscular disease: Duchenne muscular dystrophy. Am J Phys Med Rehabil 1995: 74(2):S70-92.
- Emery AH, Population frequencies of inherited neuromuscular diseases – a world survey. Neuromusc Disord 1991; 1:19-29.
- McDonald CM, Abresch RT, Carter GT, Fowler WM, Johnson ER, Kilmer DD. Profiles of neuromuscular disease: Becker muscular dystrophy. Am J Phys Med Rehabil 1995; 74(2) S93-103.
- ER Johnson, Carter GT, Kilmer DD, Abresch RT, Fowler WM, Wanlass RL. Profiles of neuromuscular disease: myotonic muscular dystrophy. Am J Phys Med Rehabil 1995; 74(2) S104-16
- Fu YH, Friedman DL, Richards S. et al. Decreased expression of myotonin-protein kinase messenger RNA and protein in adult form of myotonic dystrophy. Science 1993; 260:235-38.
- Kilmer DD, Abresch RT, Aitkens SG, Carter GT, Fowler WM, Johnson ER, McDonald C1M. Profiles of neuromuscular disease: facioscapulohumeral dystrophy. Am J Phys Med Rehabil 1995; 74(2):S131-39.
- McDonald CM, Abresch RT, Carter GT, Fowler WM, Johnson ER, Kilmer DD, Wright NC. Profiles of neuromuscular disease limb-girdle syndromes. Am J Phys Med Rehabil 1995; 74(2):S117-30.
- Matsumura K, Campbell KP, Dystrophin-glycoprotein complex: Its role in the molecular pathogenesis of muscular dystrophies. Muscle Nerve 1994; 17:2-15.
- Matsumura K, Tome FMS, Collin H, et al. Deficiency of the 50K dystrophin-associated glycoprotein in severe childhood autosomal recessive muscular dystrophy. Nature 1992; 359: 320-22.
- Carter GT, Abresch RT, Fowler WM, Johnson ER, Kilmer DD, McDonald CM, Wright NC. Profiles of neuromuscular disease: hereditary motor and sensory neuropathy, types I and 11. Am J Phys Med Rehabil 1995; 74(2):S140-49.
- Chance PF, Alderson MK, Leppig KA, et al. DNA deletion associated with hereditary neuropathy with liability to pressure palsies. Cell 1993: 72:143-51.
- Chance PF, Matsunami N, Lensch W, Smith B, Bird TD. Analysis of the DNA duplication 17pl1.2 in Charcot-Marie-Tooth neuropathy type I pedigrees: additional evidence for a third autosomal CMT locus. Neurology 1992;42:2037-41.
- Ionasescu VV. Charcot-Marie-Tooth neuropathies from clinical description to molecular genetics. Muscle Nerve 1995; 18:267-75.
- Carter GT, Abresch RT, Fowler WM, Johnson ER, Kilmer DD, McDonald CM. Profiles of neuromuscular disease: spinal muscular atrophy. Am J Phys Med Rehabil 1995; 74(2): S150-59.
- Mackenzie AE, Jacob P, Surh L, Besner A. Genetic heterogeneity in spinal muscular atrophy: a linkage analysis-based assessment. Neurology 1994; 44:919-24.
- Eisen A. Amyotrophic lateral sclerosis is a multifactorial disease. Muscle Nerve 1995; 18 (7):741-52.
- Carter GT. Neuromuscular disorders In: Dell Orto AE, Marinelli RP (eds.) Encyclopedia of disability and rehabilitation. New York Simon & Schuster Macmillan. 1995: 509-15.
- Norris F, Sheperd R, Denys E, Mukai E, Elia L, Holden D, Norris H. Onset, natural history and outcome in idiopathic adult motor neuron disease J Neurol Sci 1993; 118 (1):48-55.
- Ringel SP, Murphy JR, Alderson MK, Bryan W, England JD, Miller RG, et al. The natural history of amyotrophic lateral scle rosis. Neurology 1993; 43 (7):1316-22.
- Tysnes OB, Vollset SE, Larsen JP, Aarli JA. Prognostic factors and survival in amyotrophic lateral sclerosis. Neuroepidemiology 1994: 13(5): 226-35.
- Delateur BJ, Giaconi RM. Effect on maximal strength of submaximal exercise in Duchenne muscular dystrophy. Am J Phys Med 1979; 58: 26-36.
- Aitkens SG, McCrory MA, Kilmer DD, Bernauer EM. Moderate resistance exercise program: its effects in slowly progressive neu romuscular disease. Arch Phys Med Rehabil 1993; 74(7):711-15.
- Kilmer DD, McCrory MA, Wright NC, Aitkins SG, Bernauer EM. The effect of a high resistance exercise program in slowly pro gressive neuromuscular disease. Arch Phys Med Rehabil 1994; 75(5):560-63.
- Lindemen E, Leffers P, Spaans F, Drukker J, Reulen J, Kerckhoffs M, Koke A. Strength training in patients with myotonic dystrophy and hereditary motor and sensory neuropathy: a randomized clinical trial. Arch Phys Med Rehabil 1995; 76(7):612-20.
- Carter GT, Kikuchi N, Horasek S, Walsh SA. The use of fluorescent dextrans as a marker of sarcolemmal injury. Histo Histopathol 1994;9(3):443-47.
- Carter GT, Wineinger MA, Walsh SA, Horasek SJ, Abresch RT, Fowler WM. Effect of voluntary wheel-running exercise on muscles of the mdx mouse. Neuromusc Disord 1995; 5(4):323-31.
- Carter GT, Kikuchi N, Abresch RT, Walsh SA, Horasek S, Fowler WM. Effects of exhaustive concentric and eccentric exercise on murine skeletal muscle. Arch Phys Med Rehabil 1994; 75(5):555-59.
- Carter GT, Longley KJ, Entrikin RK. Electromyographic and nerve conduction studies in the mdx mouse. Am J Phys Med Rehabil 1992: 71(1):2-5.
- Mitsumoto H, Norris FH. Amyotrophic lateral sclerosis: a comprehensive guide to management. New York: Demos Vermande 1994.
- Sharma KR, Kent-Braun JA, Majumdar S. Huang Y, Mynhier M, Weiner MW, Miller RG. Physiology of fatigue in amyotrophic lateral sclerosis. Neurology 1995; 45(4):733-40.
- Carter GT, Longley KJ, Walsh SA, Entrikin RK. Lack of effect of amitriptyline in murine myotonia. Am J Phys Med Rehabil 1992; 71(5):279-82.
- Aitkens S, Lord J, Bernauer E, Fowler Jr WM, Lieberman JS, Berck P. Relationship of manual muscle testing to objective strength measurements. Muscle Nerve 1989;12:173-77.
- Kilmer DD, Abresch RT, Fowler WM Jr. Serial manual muscle testing in Duchenne muscular dystrophy. Arch Phys Med Rehabil 1993;74: 1168-71.
- Lord JP, Portwood MM, Lieberman JS, Fowler WM, Berck P. Upper extremity functional rating for patients with Duchenne muscular dystrophy. Arch Phys Med Rehabil 1987; 68 151-54.
- Fowler WM, Abresch RT, Aitkens SA, Carter GT, Johnson ER, Kilmer DD, McCrory MA. Impairment and disability profiles of neuromuscular diseases: design of the protocol Am J Phys Med Rrhabil 1995; 74(2) S62-69.
- Carter GT, Kilmer DD, Szabo RM, McDonald CM. Focal posterior interosseus neuropathy in the presence of hereditary motor and sensory neuropathy, type 1. Muscle Nerve 1996; 19:644-48.
- Vignos PJ, Management of musculoskeletal complications in Neuromuscular disease:limb contractures and the role of stretching, braces and surgery. Physical Medicine and Rehabilitation :State of the Art Reviews 1988; 2:509-36.
- Bach JR, McKeon J, Orthopedic surgery and rehabilitation for prolongation of brace-free ambulation of patients with Duchenne muscular dystrophy. Am J Phys Med Rehabil 1991; 70:324-31.
- Manzur AY, Hyde SA, Rodillo E, Heckmart JZ, Bentley G, Dubowitz V. A randomized controlled trial of early surgery in Duchenne muscular dystrophy. Neuromusc Disord 1992; 2:379-87.
- Lord J, Behrman B, Varzos N, Cooper D, Lieberman JS, Fowler Jr WM. Scoliosis associated with Duchenne muscular dystrophy. Arch Phys Med Rehabil 1990; 71:13-17.
- Stern LM, Clark BE. Investigation of scoliosis in Duchenne dystrophy using computerized tomography Muscle Nerve 1988;11:775-83.
- Hsu JD, The natural history of spine curvature progression in the nonambulatory Duchenne muscular dystrophy patient. Spine 1983; 8:771-75.
- Cambridge W, Dennan JC. Scoliosis asociated with Duchenne muscular dystrophy. J Pediatr Orthop 1987; 7:436-40.
- Pecak F, Trontelj JV, Dimitrijevic MR. Scoliosis in neuromuscular disorders. Int Orthop 1980; 3:323-28.
- Smith AD. Koreska J, Eng P, Moseley CF. Progression of scoliosis in Duchenne muscular dystrophy. J Bone Joint Surg 1989; 71A:1066-74.
- Johnson EW, Yarnell SK. Hand dominance and scoliosis in Duchenne muscular dystrophy. Arch Phys Med Rehabil 1976; 57:462-64.
- Seeger BR, Sutherland AD, Clark MS, Orthotic management of scoliosis in Duchenne muscular dystrophy. Arch Phys Med Rehabil 1984; 65:83-86.
- Colbert AP, Craig C. Scoliosis management in Duchenne muscular dystrophy: Prospective study of Modified Jewett hyperextension brace. Arch Phys Med Rehabil 1987; 68:302-304.
- Oda T, Shimizu N, Yonenobu K, Ono K, Nabeshima T, Kyoh S. Longitudinal study of spinal deformity in Duchenne muscular dystrophy. J Pediatr Orthop 1993; 13:478-88.
- Rodillo EB, Fernandez-Bermejo E, Heckmatt JZ, Dubowitz VA. Prevention of rapidly progressive scoliosis in Duchenne muscular dystrophy by prolongation of walking with orthoses. J Child Neurol 1988; 3:269-74.
- Sakai DN, Hsu JD, Bonnett CA, Brown JC. Stabilization of the collapsing spine in Duchenne muscular dystrophy. Clin Orthop 1977; 128:256-60.
- Millr RG, Chalmers AC, Dao H, Filler Katz A, Holman D, Bost F. The effect of spine fusion on respiratory function in Duchenne muscular dystrophy. Neurology 1991; 41:38-40.
- Jenkins JG, Bohn D, Edmonds JF, Levison H, Barker GA. Evaluation of pulmonary function in muscular dystrophy patients requiring spinal surgery. Crit Care Med 1982; 10:645-49.
- Kurz LT, Mubarak SJ, Schultz P, Min Park S, Leach J. Correlation of scoliosis and pulmonary function in Duchenne muscular dystrophy. J Pediatr Orthop 1983; 3:347-53.
- Griggs RC, Donohoe KM, Utell MJ, Goldblatt D, and Moxley R. Evaluation of pulmonary function in neuromuscular disease. Arch Neurol 1981; 38:9-12.
- Bach JR, Amyotrophic lateral sclerosis: predictors for prolongation of life by noninvasive respiratory aids. Arch Phys Med Rehabil 1995:76 (9):828-32.
- Rideau Y, Jankowski LW, Grellet J. Respiratory function in the muscular dystrophies. Muscle Nerve 1981; 4:155-64.
- Fukunaga H, Okubo R, Moritoyo T, Kawashima N, Osame M. Long-term follow up of patients with Duchenne muscular dystrophy receiving ventilatory support. Muscle Nerve 1993; 16: 554-58.
- Bach JR. Amyotrophic lateral sclerosis: communication status and survival with ventilatory support. Arch Phys Med Rehabil 1993; 72(6):343-49.
- Carter GT, Kilmer DD, Bonekat HW, Lieberman JS, Fowler WM. Evaluation of phrenic nerve and pulmonary function in hereditary motor and sensory neuropathy, type 1. Muscle Nerve 1992: 15:459-62.
- Carter GT. Phrenic nerve involvement in Charcot-Marie-Tooth. Muscle Nerve 1995; 18:1215-16.
- Strand EA, Miller RM, Yorkston KM, Hillel AD. Management of oral-pharyngeal dysphagia symptoms in amyotrophic lateral sclerosis. Dysphagia 1996; 11:129-39.
- Carver GT, Johnson ER, Bonekat HW, Lieberman JS. Larvngeai diversion in the treatment of intractable aspiration in motor neuron disease. Arch Phys Med Rehabil 1992; 73(7):680-82.
- D’Orsogna LD, O’Shea JP, Miller G. Cardiomyopathy of Duchenne muscular dystrophy. Pediatr Cardiol 1988: 9:205-13.
- Backman E, Nylander E. The heart in Duchenne muscular dystrophy: a non-invasive longitudinal study. Eur Heart J 1992: 13:1239-44.
- Bies RD, Friedman D, Roberts R, Perryman MB, Caskey CT. Expression and localization of dystrophin in human cardial Purkinje fibers. Circulation 1992; 86:147-53.
- Kawashima S, Ueno M, Kondo T, Yamamoto J, Iwasaki T. Marked cardiac involvement in limb-girdle muscular dystrophy. Am J Med Sci 1990; 299:411-14.
- Willig TN, Carlier L, Legrand M, Riviere H, Navarro J. Nutritional assessment in Duchenne muscular dystrophy. Dev Med Child Neurol 1993: 35:1074-82.
- Jaffe KM, McDonald CM, Ingman E, Hass J. Symptoms of upper gastrointestinal dysfunction: Case-control study. Arch Phys Med Rehabil 1990; 71:742-44.
- Marsh GG, Munsat TL. Evidence of early impairment of verbal intelligence in Duchenne muscular dystrophy. Arch Dis Child 1974; 49:118-22.
- Liu J, Lissens W, Van Broeckhoven C, Lofgren A, Camus M, Liebaers I, Van Steirteghem A. Normal pregnancy after preimplantation DNA diagnosis of a dystrophin gene deletion. Prenat Diagn 1995; 15(4):351-58.
- Bensimon G, LaComblez L, Meininger V, ALS/Riluzole Study Group. A controlled trial of riluzole in amyotrophic lateral sclerosis. N Engl J Med 1994; 330:585-91.
- Miller RG, Petajan JH, Bryan WW, Armon C, Barohn RJ, Goodpasture JC, et al. A placebo-controlled trial of recombinant human ciliary neurotrophic (rhCNTF ) factor in amyotrophic lateral sclerosis (ALS). Ann Neurol 1996: 39:256-60.
- Backman E, Henriksson KG. Low dose prednisolone treatment in Duchenne and Becker muscular dystrophy. Neuromuscul Disord 1995; 5(3):233-41.
- Brooke MH, Griggs RC. Mendell JR, Fenichel GM, Shumate JB, Pellegrino RJ. Clinical trial in Duchenne dystrophy.1. The design of the protocol. Muscle Nerve 1981:4:186-97.
- Carter GT, Bonekat HW, Milio L. Successful pregnancies in the presence of spinal muscular atrophy: two case reports. Arch phys Med Rehabil 1994: 75(2):229-31
Management of Myotonic dystrophy
This information is from the 11th annual conference of the English Myotonic Dystrophy
“Myotonic Dystrophy Incurable but not Unmanageable”
The incidence of Myotonic Dystrophy is 1 in 8000. Most general practioners have about 2000 patients in their practice. So by simple mathematics only one in four general practioners will see a patient with DM. Most will not know much about it. So it is necessary to have specialized centers for treatment. (Webmaster note: By implication the congenital form is about 1 in 100,000 so that only 1 in 50 pediatricians will have a patient with CMyD in their practice)
Most other muscular dystrophies will only affect the muscles. DM is different in that other systems are affected as well. Another difference is that with most other genetic diseases you either have the gene and the disease or you don’t. DM is a variable genetic disease. It severity differs with the age of onset and the number of repeats. There is not a distinct expression of the disease based on an on or off view. Thus, many medical practioners will have a harder time understanding the disease. The other implication is that you can not generalize the condition of one patient with the disease to another, it is variable
Skeletal Muscle: The skeletal muscle is defined by Myotonia of muscular stiffness, in particular hand stiffness. Also certain other muscles are weak especially facial, scapula and humorous muscles are mostly affected. However, this stiffness is generally not he problems that patients report more, it is the weakness of the hand and other muscles that comes into play. This is more the day to day problems that weaker muscles may cause. Weakness of the grip seems to be the most problematic. A normal male should be able to squeeze about an equivalent of 50Kg. A patient with DM may only be able to squeeze the equivalent of 10-1Kg for example. Also weakness of the ankle muscles causes the characteristic gait of DM. Dr. Jones mentioned that a patient of his was unable to blow into a breathalyzer machine So Dr. Jones felt it was more the practical issues of weakness more than the myotonia that affect DM patients
Smooth Muscles: One of the primary effects of DM on smooth muscle is that swallowing and feeding are much more difficult. What is generally recommended is that food should be cut into small pieces and drink used to wash this down. Care must be taken to avoid aspiration pneumonia, that is food going down into the lungs and causing pneumonia. Dr, Jones mentioned a drug Cysotia?? (Margaret can we get clarification here) or surgery that might be needed to assist. Also a video Fluoroscopy will help diagnosis any feeding problems.
The smooth muscles also affects the bowel and notably irritable bowel syndrome is very common in patients with DM. This is failure of coordination of muscles. Constipation is also a complication because of muscle problems plus immobility of patients not moving. Surgery is not recommend and Dr. Jones has heard reports of patient deaths with surgery. The Uterus is also affected and problems in childbirth may result.
The heart is affected in other dystrophies but in DM it is not the heart muscle itself that is affected. Rather, it is problems with the electrical signals not working properly and the heart either working to fast or too slowly. Atrial fibrillation is when part of the heart is not working efficiently, pumping to fast and not coordinated) and this can cause dizzy spells and blackouts, can be treated. The heart can also pump too slow causing the same symptoms. Heart changes are slow to respond to changes in the body. Dr. Jones recommended an EKG every year to look for problems.
Eyes and Ears: Cataracts can be the first evidence of myotonic dystrophy disease. Generally, if cataracts are seen in patients under 50 DM should be considered. There is nothing special about the mature form of cataracts. However, in the early stages there is a characteristic pattern that can be seen that is unique to DM. A mild degree of deafness is due to DM but not serious.
Brain. On average IQ is a little lower. In the congenital form IQ is reduced and most children will have learning disabilities. The mental problems are pretty static with little change over time.
Excessive Daytime Sleeping: 75% of DM patients have excessive sleepiness. In a survey they conducted 120 responses were received and 40% of patients had a pathological sleeping problem. There is a new drug Modafinil (Brand name Provigil in USA) that might help. Make sure to take it in the morning!
Breathing: The lungs are affected as well as the heart. The anesthesiologist should be informed as well as the surgeon prior to any surgery. Certain precautions must be taken. DM suffers should wear a bracelet as well as cards in case of an accident. The group had a handout on anesthesia that was in some of there earlier newsletters.
Endocrine System: There is reduced fertility in patients with DM. There is also diabetes. Dr. Jones didn’t feel that DM diabetes falls into the classical sense of Diabetes. Feels that all patients with DM have problems with sugar levels. (My note: Might possibly have a different Mechanism of action?). Would be nice to get some more information on this.
Dr. Jones feels that the age of onset is a more potent predictor of outcome versus the number of repeats. There is the anticipation factor where in each generation the outcome becomes progressively worse.
Genetic counseling: Family needs to be screened especially women to prevent them from unknowingly having an infant with CMyD. When an individual is identified strong efforts should be considered to have other family members tested to avoid them having CMyD. For example Dr. Jones considered an example of a 28 year old male who was confirmed diagnosis with DM. He had 2 sisters one 25 and one 21. The 21 year old sister went on to have a child with CMyD because she was not informed or tested.
Link to Article by Dr. Carter on Management of Neuromuscular Diseases

